Scientists Have Eliminated Pancreatic Cancer in Mice Without Major Side Effects
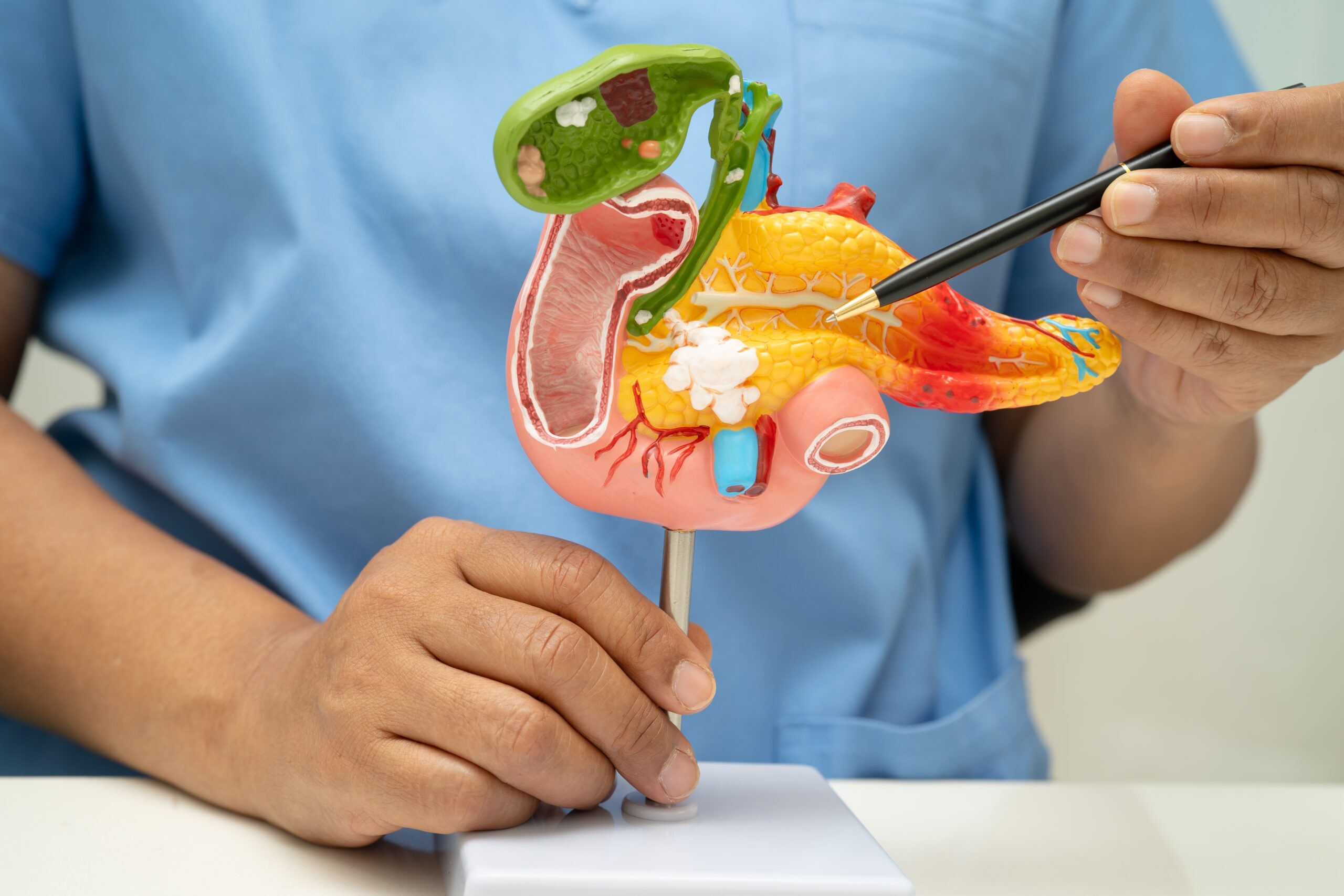
Pancreatic cancer has long stood as an unbreachable wall in modern medicine, with survival rates that have barely budged in fifty years. It is a diagnosis that typically comes with a sense of finality, resisting standard treatments with a relentless adaptability.
Yet, a groundbreaking shift is happening in a laboratory in Spain. Researchers have not just slowed the disease in animal models—they have completely eliminated it, suggesting that even the most aggressive tumors may finally have a fatal weakness.
Breaking the Cycle of Resistance
Pancreatic cancer notoriously resists treatment. Standard therapies often fail because the tumors adapt and survive, finding new routes to grow even when one path is blocked. This adaptability is why the five-year survival rate remains below 10 percent. However, a groundbreaking study from the Spanish National Cancer Research Centre (CNIO) has demonstrated a potential way to dismantle these defenses completely.
Led by Dr. Mariano Barbacid, a pivotal figure in cancer genetics, researchers successfully eliminated aggressive pancreatic tumors in laboratory mice. They achieved this not by relying on a single drug, but by deploying a “triple-drug” therapy. This strategy targets the KRAS pathway, a genetic mutation found in 90 percent of pancreatic cancer cases, at three separate points simultaneously.
Think of the cancer’s survival mechanism like a bridge. If you cut one support cable, the bridge might hold. If you cut three critical cables at once, the structure collapses. By blocking the KRAS pathway at multiple stages, the therapy prevented the cancer cells from adapting. The results published in the Proceedings of the National Academy of Sciences (PNAS) were stark. The tumors did not just shrink; they disappeared. Furthermore, the mice remained cancer-free during extended follow-up periods without suffering significant side effects. This marks the first time such complete and durable regression has been achieved in these specific experimental models.
Forty Years in the Making

This breakthrough is the result of decades of specialized focus. Dr. Mariano Barbacid is a giant in the field, having played a pivotal role in identifying the first human oncogene in the early 1980s. This discovery fundamentally changed how the world understands cancer as a genetic disease. For the last forty years, his research has centered on KRAS-driven tumors, which are notoriously difficult to treat. Barbacid has long argued that a single-drug approach is insufficient because of the disease’s extraordinary ability to adapt and survive.
Current treatments affirm his theory. While the first drugs targeting KRAS mutations were approved in 2021, their success is often short-lived. The tumors develop resistance within months, rendering the medication ineffective. To overcome this biological deadlock, the team at the Spanish National Cancer Research Centre employed a specific chemical arsenal.
They utilized a combination of three distinct agents: daraxonrasib, an experimental KRAS inhibitor; afatinib, a drug already approved and used for lung cancer; and SD36, a protein degrader. This was not a random mixture but a calculated siege on the cancer’s communication network. By integrating a drug already in clinical use with advanced experimental compounds, the study demonstrates that the tools to fight this disease may already exist or are within reach. It requires the right combination to unlock their full potential and stop the tumor from mutating its way out of extinction.
A Blueprint for Future Success

While the elimination of tumors in animal models is historic, a crucial distinction remains. Mice are not humans. The biological gap between the two is where many promising treatments fail. Dr. Barbacid and his team are transparent about this limitation. He emphasizes that they are not yet in a position to launch clinical trials with this specific triple therapy.
The immediate challenge is toxicity. The dosage and combination that saved the mice must be rigorously optimized for human biology. The goal is to ensure the treatment remains lethal to the cancer but safe for the patient. This process is complex and will take time. There is no overnight miracle here.
However, this caution does not diminish the significance of the findings. The study provides a confirmed proof of concept. It proves that pancreatic ductal adenocarcinoma, often viewed as invincible, has vulnerabilities. The path forward involves refining these chemical agents to bridge the divide between laboratory success and clinical application. This is not the end of the road, but for the first time in decades, researchers have a clear map of where to go next.
Rewriting the Prognosis
For decades, a diagnosis of pancreatic ductal adenocarcinoma was effectively a final verdict. The numbers paint a grim picture. In Spain alone, over 10,300 cases arise annually, and fewer than 10 percent of patients survive past five years. Standard chemotherapy has stalled, and conventional treatments often hit a wall because they fail to address the genetic complexity of the tumor.
This study represents a crucial pivot. It was not an accident but the product of rigorous, funded investigation supported by organizations like the CRIS Cancer Foundation and the European Research Council. The involvement of these major institutions underscores a vital truth in medical science: breakthroughs require sustained resources and collective effort.
The narrative is finally shifting. We are witnessing the move from hopelessness to strategy. By proving that resistance can be beaten in the lab, these scientists have validated that the disease is not invincible. It requires the right resources, the right strategy, and the persistence to dismantle the problem piece by piece. What was once deemed impossible is now a matter of engineering the right solution.
A Matter of When, Not If
Progress is rarely a straight line. It is a series of failures, adjustments, and small victories that eventually change the world. This breakthrough serves as a powerful reminder that “incurable” is often just a temporary label waiting for the right solution.
For forty years, experts analyzed the genetic armor of pancreatic cancer. They did not stop when single drugs failed. They did not stop when the statistics remained bleak. They adapted. This adaptability is the greatest tool humanity possesses.
Do not mistake a long road for a dead end. While this therapy is not yet in hospitals, it represents a definitive crack in the armor of a terrifying disease. It validates the necessity of funding rigorous, long-term research rather than seeking instant gratification.
For patients and families navigating this diagnosis, let this be a source of strength. The brightest minds are closing in on the answer. Science is moving from understanding the problem to engineering the solution. The fight is difficult, and the waiting is painful, but the direction is finally clear. The solution is no longer a question of if, but when.
Featured Image Source: Shutterstock
Source:
- Liaki, V., Barrambana, S., Kostopoulou, M., Lechuga, C. G., Zamorano-Dominguez, E., Acosta, D., Morales-Cacho, L., Álvarez, R., Sun, P., Rosas-Perez, B., Barrero, R., Jiménez-Parrado, S., López-García, A., Roman, M. S., López-Gil, J. C., Drosten, M., Sainz, B., Musteanu, M., Caleiras, E., . . . Barbacid, M. (2025). A targeted combination therapy achieves effective pancreatic cancer regression and prevents tumor resistance. Proceedings of the National Academy of Sciences, 122(49), e2523039122. https://doi.org/10.1073/pnas.2523039122
Loading...

