Doctors Have Been Treating Kidney Stones for Centuries. They Just Found Something Living Inside Them.
Something was hiding inside the kidney stones. Something nobody had thought to look for. Something that, once found, made researchers stop and reconsider decades of medical thinking.
Scientists had studied kidney stones for centuries. They had mapped their mineral composition, tracked their formation, and developed treatments to break them apart or flush them out. Doctors told patients to drink more water, change their diet, and come back if the pain returned. Many patients did come back. Again and again. And for a long time, nobody could fully explain why.
A new study published in January 2026 in the Proceedings of the National Academy of Sciences may have finally uncovered part of that answer. What researchers found inside the most common type of kidney stone has quietly rewritten one of medicine’s oldest assumptions.
What We Thought We Knew
Kidney stones form when minerals in urine crystallize and clump together into hard deposits inside the kidneys. About 1 in 11 people will develop them at some point in their lives, and that number has been climbing globally for decades.
Calcium oxalate stones make up nearly 80% of all cases. For as long as medicine has studied them, doctors classified these stones as purely chemical events. Minerals build up. Crystals form. A stone grows. No living organisms involved.
One rare type of kidney stone, called a struvite stone, had long been linked to bacterial infection. Struvite stones account for only 2 to 6% of cases, and doctors had always treated them differently because of that bacterial connection. Calcium stones, by contrast, carried the label “noninfectious.” Clean. Abiotic. Nothing living inside them. That label just got pulled off the shelf.
What the Researchers Actually Found

A UCLA-led team set out to study calcium kidney stones using advanced microscopy techniques. What they expected to find was a straightforward mineral structure. What they found instead stopped them cold. Live bacteria. Layers of bacterial biofilm. Embedded not on the surface of the stones, but deep within their internal structure.
“This breakthrough challenges the long-held assumption that these stones develop solely through chemical and physical processes, and instead shows that bacteria can reside inside stones and may actively contribute to their formation,” said Dr. Kymora Scotland, an assistant professor of urology at the David Geffen School of Medicine at UCLA and the study’s co-senior author.
Bacteria were not just sitting on the outside of these stones like hitchhikers. They were woven into the architecture of the stone itself, sandwiched between layers of crystal as if they had always belonged there. When researchers looked at polished cross-sections of calcium stones under a microscope, they saw alternating striations, dark bands rich in bacterial material and lighter bands of mineral crystal, layered on top of each other like rings inside a tree trunk. Every calcium oxalate stone fragment evaluated showed the same pattern.
How Bacteria Get Locked Inside a Stone
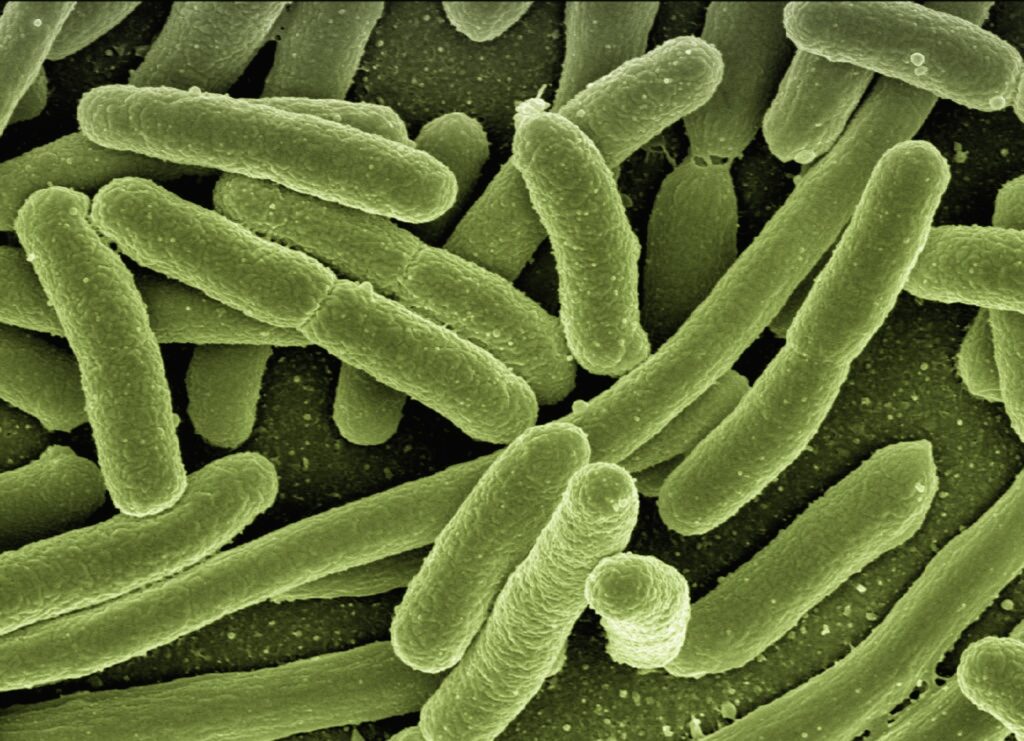
Here is where it gets genuinely strange. Bacteria may not just be passengers caught inside a forming stone. They may be helping build it. When bacteria live in groups, they produce a protective layer called a biofilm, a sticky matrix of proteins, sugars, and DNA that holds the colony together and shields it from outside threats. Inside that biofilm, bacteria release what scientists call extracellular DNA, genetic material that spills out into the surrounding environment.
DNA carries a strong negative electrical charge. Calcium ions carry a positive charge. Put them together in the mineral-rich environment of the kidney, and something predictable happens. Calcium ions get pulled toward the DNA. They cluster around it. And those clusters become the starting points for crystal growth.
In other words, bacteria release DNA. DNA attracts calcium. Calcium crystallizes. A stone begins to form around the bacteria. As more layers grow, the bacteria get locked inside. Layer by layer, crystal by crystal, the stone seals them in.
Researchers found that crystals near bacterial layers were noticeably smaller than crystals elsewhere in the stone. Smaller crystals mean more starting points for growth, exactly what you would expect if bacteria were seeding the process from within.
Why Doctors Keep Missing It

Here is the part that should make every kidney stone patient pay attention. Standard clinical tests often failed to detect any bacteria in these stones. Researchers found bacteria in 24 out of 54 stones tested, including many that had returned negative results on conventional culture tests. Of the stones analyzed using more targeted methods, 17 out of 22 contained culturable bacteria. Over 30% showed multiple bacterial species living together inside a single stone.
How does a stone full of bacteria test negative for bacteria? Most bacteria do not behave like the strains scientists grow in labs. An estimated 99% of bacteria in nature cannot survive standard laboratory culturing conditions. Inside a kidney stone, bacteria can enter a low-activity state where they are technically alive but not actively growing. Standard tests look for growth. No growth means no bacteria detected. No bacteria detected means the stone gets labelled clean. Clean, as it turns out, was never the right word.
The Recurring Problem Nobody Could Solve

Every person who has passed a kidney stone describes it as one of the worst pains of their life. Sharp, relentless, and radiating from the lower back through the abdomen. Many patients require surgery or a procedure called lithotripsy, which uses sound waves to break stones apart from outside the body.
After treatment, doctors send patients home with instructions to stay hydrated and modify their diet. Yet recurrence rates for certain stone types reach up to 80%. Patients who follow every instruction still come back with new stones. Some come back with infections, too, urinary tract infections that seem to appear from nowhere, even in patients who had clear urine cultures before surgery.
“We found a new mechanism of stone formation that may help to explain why these stones are so common,” Scotland said. “These results may also help to explain the connections between recurrent urinary tract infections and recurrent kidney stone formation, and provide insights on potential future treatment for these conditions.”
Consider what happens during lithotripsy. Sound waves shatter a stone into fragments small enough to pass through urine. Doctors consider it a clean procedure. But if bacteria are living inside that stone, shattering it means releasing them directly into the urinary tract. Bacteria that standard tests have never detected. Bacteria that had been sitting dormant inside the stone’s mineral layers, protected from the immune system, protected from antibiotics, waiting.
When those fragments break apart, the environment changes. A bacterium that appeared nonviable in one condition may, given enough time and a suitable new environment, wake up and start growing again. Recurring infections after stone treatment. Recurring stones after dietary changes. Both may trace back to the same hidden source.
What Changes Now

Current treatment for calcium kidney stones follows a straightforward path: drink more water, cut back on salt and certain foods, take medications to reduce mineral buildup, and remove stones surgically when necessary. Antibiotics occasionally feature in treatment plans for struvite stones, but doctors rarely consider them for calcium stones because calcium stones were never thought to involve bacteria. That thinking needs to change.
Anti-biofilm therapies, treatments designed specifically to break down or prevent bacterial biofilm formation, represent a new direction that researchers believe could change outcomes for millions of patients. Rather than managing the mineral side of stone formation alone, future treatments may need to address the bacterial side at the same time.
Anti-biofilm approaches carry an advantage that broad antibiotic use does not. Antibiotics target bacteria directly and risk driving antibiotic resistance. Anti-biofilm strategies target the structure bacteria build to protect themselves, a different angle that may produce fewer resistance concerns.
Researchers are careful to note that none of this translates to clinical practice tomorrow. More studies must happen before any new treatment reaches patients. But the direction has shifted.
A Very Old Problem, Seen With New Eyes
Kidney stones have been with us for as long as recorded history. Doctors have found them in Egyptian mummies. Ancient medical texts describe them. Humans have been treating them, dreading them, and passing them in agony for thousands of years. For most of that time, medicine looked at kidney stones and saw a mineral problem. A chemistry problem. Something to dissolve, remove, or prevent through diet.
Now, looking more closely at something that has always been right in front of them, researchers have found something else entirely living inside. Something small, quiet, and possibly responsible for far more suffering than anyone realized.
One assumption, held for decades by doctors around the world, has crumbled under a microscope. Not because someone went looking for something revolutionary, but because a team sat down to do careful work and refused to ignore what they saw. Medicine tends to move slowly. But moments like this one remind us that some of its most important turns come not from bold predictions, but from paying close attention to what was already there.
Loading...

